Premalux AB
Sweden
This website uses cookies to ensure that you get the best possible experience when visiting the site.
Please see our Privacy Policy for more information. To accept the use of non-essential cookies, please click “I agree”.



Premalux AB is a Swedish company that has been involved in early-stage research focused on addressing complications associated with extremely premature birth.
The company's work contributed to foundational research efforts within neonatal medicine, which led to the development of the therapeutic candidate now known as OHB-607.
Premalux holds certain intellectual property rights related to this work, including rights to future milestone payments and royalties contingent upon potential regulatory approval of OHB-607.
The company's current role is primarily administrative, including oversight of historical agreements and ensuring compliance with contractual obligations related to its intellectual property.
All clinical development activities related to OHB-607 are conducted by external organizations.
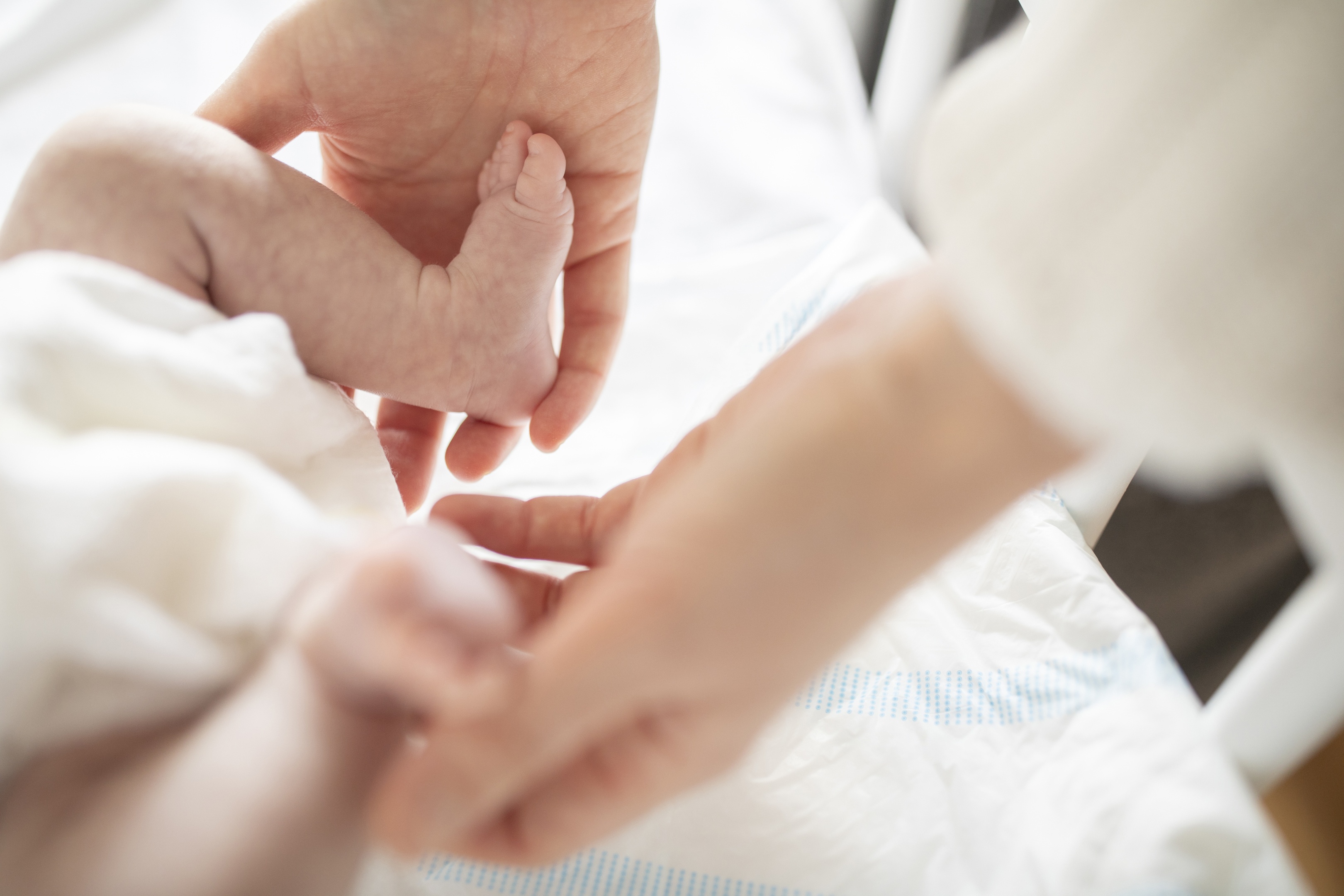
Every year, hundreds of thousands of infants worldwide are born extremely prematurely and, as a result, suffer from severe complications in their lungs, brain, and eyes — complications that can hinder long-term development and quality of life.
OHB-607 (formerly TAK-607) is a proprietary, recombinant version of insulin-like growth factor 1 (IGF-1) combined with its binding protein IGFBP-3. It represents a straightforward replacement therapy approach.
Mothers are the primary source of IGF-1 for the developing fetus, with the fetus producing very little of its own until approximately 30 weeks of gestational age. At birth, extremely premature infants — born at less than 28 weeks — have low levels of IGF-1, which are associated with greater complication rates.
OHB-607 is designed to make up for the IGF-1 deficiency that occurs in extremely preterm birth, thereby helping to promote continued development and maturation of vital organs and the vasculature that supports them.
According to publicly available information, OHB-607 has shown a statistically significant improvement in preventing severe bronchopulmonary dysplasia (BPD) and a positive trend in reducing intraventricular hemorrhage in a Phase 2a clinical trial. The therapy is now being evaluated in a Phase 2b study called Footprints, conducted by Oak Hill Bio in collaboration with Chiesi Group.
Premalux AB holds certain intellectual property rights related to the early development of this therapy, including rights to future milestone payments and royalties contingent upon potential regulatory approval. Premalux is not responsible for the ongoing clinical development or clinical trials.
For information regarding clinical trials, please refer to clinicaltrials.gov or the organizations responsible for the program.
Information that may not be shared publicly includes, but is not limited to:
confidential contractual terms
proprietary research data
unpublished clinical data
regulatory communications
internal strategy or development plans
information received under confidentiality agreements
All individuals within the Premalux organization are bound by confidentiality in the handling of sensitive information.

Premalux AB is committed to protecting your personal privacy. This website does not collect personal data beyond what is required to provide basic functionality.
Any contact inquiries sent to Premalux are handled confidentially and used solely to respond to the inquiry in question.
Premalux does not share personal data with third parties and does not store sensitive information without explicit consent.
For questions regarding how your personal data is handled, please contact us at jacob@testor.se.

There is currently no notice of a general meeting. Information regarding upcoming general meetings will be published here
